It is widely used in photoelectric cells.It is stored under a liquid, such as paraffin, which does not react.It comes in the form of a soft solid that is easy to cut.It is the 5th lightest and lightest metal.It is a soft solid in its natural form, which cuts easily.It oxidizes very quickly when exposed to air.Its melting point is 65.5 degrees Celsius and its boiling point is 759 degrees Celsius. 
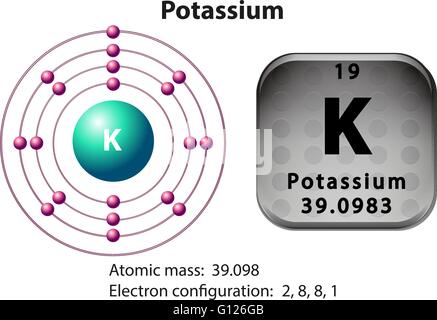
It reacts violently on contact with water, in which case it produces hydrogen.And the electron configuration of potassium is 1s2 2s2 2p6 3s2 3p6 4s1. Hence the electron arrangement in potassium is 2, 8, 8, 1. It is identified by the letter K in the periodic table and by the atomic number 19. Now the atomic number of potassium (K) is 19.It has an average radius of 220 pm, a covalent radius of 196 pm, and an atomic or Bohr radius of 243 pm.The 40K isotope, which has a half-life of about 1.278 times 109, eventually decays into the 40Ar isotope.īy using this disintegration as a method of dating rocks, we know the approximate age of certain minerals such as niotite, volcanic feldspar, muscovite, etc. Potassium has a total of 16 isotopes, three of which are naturally occurring: 39K, 40K, and 41K. It has a melting point of 336.53 degrees Kelvin and a boiling point of around 1032 degrees K. Potassium has a very low ionization energy and very low electron affinity. This element is part of the alkali metals, which are distinguished because they have only one electron in their outermost energy level, which is usually lost, thus creating a mono-positive ion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
